| Physics 320 |
Astrophysics I: Lecture #14
|
Prof. Dale E. Gary
NJIT
|
Mercury
Surface Temperatures of Planets
Before we begin to discuss Mercury, let us take a look at the general situation with regard to surface temperatures of planets, relative to the temperature of the central star. An important property of
a planet is its albedo. This
is the fraction of sunlight reflected from the surface, relative to the
amount striking the surface:
A = amount reflected / amount incident
A perfectly reflective planet would have an Albedo of 1, while a completely black planet would have an albedo of 0. Wikipedia lists the albedo
of the planets, where you can see that it varies from very dark (0.11 for
the Moon) to very bright (0.67 for cloud-covered Venus). The gas giants
have albedos near 0.5. Earth's albedo is highly variable (due to
clouds), but averages around 0.37. Saturn's moon Enceladus has an albedo above 1 (1.4)! Keep in mind that albedo numbers are difficult to calculate, and vary quite a bit in different references.
We can measure the
amount of light reflected from a planet, but how do we know the amount
striking it? The light, of course, originates at the Sun, which radiates
as a blackbody of temperature about
T
= 5800 K. The blackbody spectrum peaks at a wavelength that depends
only on temperature:
lmax
= 2.898 x 10-3
m/T
a relationship called Wien's
Law. The Sun's spectrum peaks around 500 nm. The
spectrum of an object at room temperature (about 290 K) peaks in the infrared
(about 10 mm).
Thus, we can tell the surface temperature of a planet by measuring its
blackbody spectrum. A blackbody spectrum has a total energy flux
F
(energy per unit area per unit time) given by the Stefan-Boltzmann
Law
F = sT 4
W/m2,
where s = 5.67 x 10-8
W m-2 K-4.
Since we know the energy flux
from the Sun, we can calculate the blackbody temperature one would expect
at the surface of a planet from its distance from the Sun, and its albedo
A.
It should be obvious that the energy flux from the Sun integrated over an entire sphere centered on the Sun (called the Luminosity) is the same no matter how big the sphere is. Therefore, 4pR2sunsT 4sun = 4pr2psT 4p, where rp is the distance of the planet from the Sun, and Tp is the corresponding temperature at that distance if all of the energy were absorbed by the planet (A = 0). If some of the energy is reflected (A > 0), then the temperature of the planet will be less than this. We can take this into account by including a factor (1 - A), so the relation becomes 4pR2sunsT 4sun = 4pr2p (1 - A) sT 4p. Solving for the sub-solar temperature of the planet, we have
Tss =
394 (1 - A)1/4(rp)-1/2
K, (rp= distance from
Sun to planet, in AU).
For a planet that rotates slowly,
or has little atmosphere, the sub-solar temperature is the relevant one
to use. However, when the planet is rotating rapidly, or has a substantial
atmosphere, the redistribution of energy must be taken into account.
When this is done, one gets the equilibrium
blackbody temperature:
Teq =
279 (1 - A)1/4(rp)-1/2
K, (rp= distance from Sun
to planet, in AU).
In most cases these match the appropriate temperature
calculated above, but Venus and Jupiter disagree. Venus has extra
heating due to the greenhouse effect, while Jupiter has some internal heat
source. We will discuss both of these later.
So for Mercury, which is at rp = 0.387 AU (semi-major axis), and has a very low Albedo of about 0.142, its sub-solar temperature is predicted to be about 605 K, while its equilibrium temperature would be about 431 K. These agree pretty well with accepted values of maximum temperature at the equator (about 700 K) and average temperature (340 K), keeping in mind that Mercury has a rather high eccentricity of 0.206, and it spends more than half of its time farther away that 0.387 AU. However, on the dark side of Mercury the equator temperature can be as low as 100 K (-173 C, or -279 F).
Dating Rocks
We can date the Earth by using radioactive decay of atoms. Atoms come in many isotopes, all having the same number of protons, but different numbers of neutrons. When an isotope has a lot of neutrons, it can be stable over a long period of time, billions of years, but still have some probability to decay to a more stable isotope. An important example is 238U (a Uranium isotope with 238 nucleons--protons+neutrons), which decays to 206Pb with a half-life of 4.5 billion years. This means that 1/2 of a sample of 238U will decay into 206Pb in 4.5 billion years. Thus, one can take a sample of rock and compare the ratio of these two isotopes to get the age of the rock. This works because 206Pb is a rare isotope in nature, so one can be quite sure that the rock had essentially none when it was first created. Another pair of isotopes that is important for dating rocks is 40K, which decays into the inert noble gas 40Ar with a half-life of 1.3 billion years. When a rock is remelted, the Ar escapes and the clock is reset, so the age one measures is the age since the last remelting of the rock.
When atoms decay, the change in the number is proportional to the number that exist (i.e., some fraction decay in a time dt). The fraction is given by a constant of proportionality l. The governing equation is
dn = -ln dt
which can be rewritten as:
dn/n = -l dt.
The integral of this equation is
ln n = -lt + constant
where the constant can be found by considering the initial conditions at time t = 0. The solution is that the rock sample starts out a number n0 at t = 0, so the solution is:
n = n0e-lt.
Now the half-life occurs when n = n0/2 at time t = t, so
t = (ln 2)/l = 0.693/l.
Let's do an example. A rock has a ratio 40Ar to 40K of 1 to 4. How old is the rock, given that the half-life is 1.3 billion years? This means that 1 out of 5 40K atoms have decayed to 40Ar, so n = n0 = 4/5. Our equation then becomes:
4/5 = e-0.693 t / t.
Solving for t, we get t = 0.322 t = 420 million years.
Mercury
Physical characteristics: One solar day is 2 Mercury years long! One sidereal day is 58.7 days, while one year is 88 days (2:3 ratio), so the synoptic rotation period is found by:
1/S = 1/P - 1/T = 1/58.7d - 1/88.0d = 0.00567 / day
so S = 176 days. Mercury's bulk density is 5420 kg/m3, about the same as for Earth, but this turns out to be a puzzle. Mercury is much less massive than Earth, so its gravity cannot squeeze rock to high density. Thus, it must be made of naturally high density material--metals. We infer a large metallic core (about 75% of the radius) covered with a rocky mantle. One possibility is that Mercury was once much larger, but suffered a massive collision that made it lose most of its outer mantel (Wikipedia article, Messenger link).
The albedo is quite low, at 0.142, giving a subsolar temperature of a whopping
Tss = 394 (1-A)1/4 (rp)-1/2 = 394 (0.96) (0.307)-1/2 = 689 K (at perihelion).
Yet during the long solar night, the dark-side temperatures drop to 100 K.
Surface Features:
The Mercury Messenger spacecarft arrived at Mercury in March 2011, and is now busy making the most detailed maps and analysis of the planet ever. As you know, going to Mercury is energetically expensive! The Messenger mission had to use several planetary encounters (with Earth, Venus, and Mercury) to reduce speed enough to get into Mercury orbit. The Messenger mission has 7 key questions it is trying to answer.
An earlier flyby mission, Mariner, 10 discovered a very weak, but present, magnetic field of 300 nT (nanotesla). Earth's magnetic field is some 40000 nT, more than 100 times larger than Mercury. Still, this is large enough to deflect the solar magnetic field and solar wind, creating a small magnetosphere. First results just released from Messenger mapped the magnetic field and found the surprising result that its center is offset some 20% of Mercury's diameter toward the north. It was not clear until recently where such a magnetic field would come from, since Mercury rotates slowly, but recently it has been discovered that it has a liquid outer core, like Earth (although much thinner).
Mercury's atmosphere, more properly called an exosphere, is very thin and consists of sodium atoms that are released from the surface by particle radiation and hang around for awhile but ultimately escape. It is of scientific interest to know the distribution of this exosphere, and in particular its dawn-dusk asymmetry, which can be observed from Earth during a transit of Mercury. A transit occurred earlier this year (2016 May 9), and was observed with NJIT's Big Bear Solar Observatory (see video). From these observations, the (preliminary) image in Figure 1a was obtained.
 |
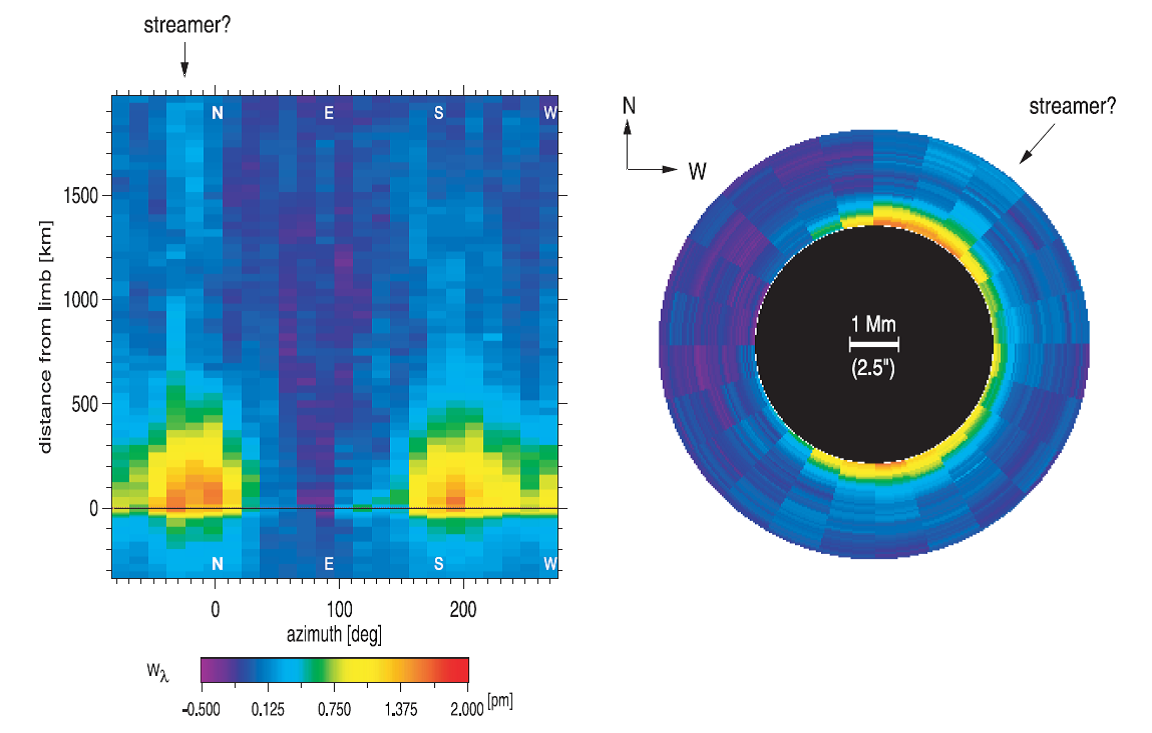 |
| Figure 1a: Preliminary data from the 2016 May 9 transit of Mercury across the Sun, taken with Big Bear Solar Observatory's FISS instrument, showing the distribution of sodium absorption in Mercury's atmosphere (dark feature extending above the planet). Carl Schmidt, private communication. |
Figure 1b: A similar observation from the 2003 Mercury transit, with somewhat lower spatial resolution, but generally agreeing with the 2016 results shown in Fig. 1a. Schleicher et al. 2004, "Detection of neutral sodium above Mercury during the transit on 2003 May 7, A&A 425, 1119. |
By taking images in many wavelengths, Messenger has made true-color and exaggerated color images. The images reveal something about the mineral makeup of the surface, as do true-color images like this one of the crater Degas.
BepiColombo mission (ESA)

